In the last years of the 19th century, scientists began to think about obtaining materials that could replace wood, ivory or mother-of-pearl. Using natural macromolecular substances, the first polymers and plastics were created.
Nature itself has come up with not a small number of such rare compounds. First of all, protein polymers, rubber compound, polysaccharide and nucleic acid. After a few years, the need to use organic compounds disappeared, since they learned how to synthesize phenol-formaldehyde resins. On their basis, materials are extracted that are similar in characteristics to wood, metal and other durable alloy. Only in the compounds obtained properties are improved.
Why are they needed?
Using artificially created substances, a large number of plastic products are created. The reason for this was the ability at the molecular level to rebuild and take on a new form, conceived by the manufacturer.
Asbestos is in demand in construction. Manufacture of slate and all kinds of pipes. In the textile industry, fire-resistant suits are sewn from it.
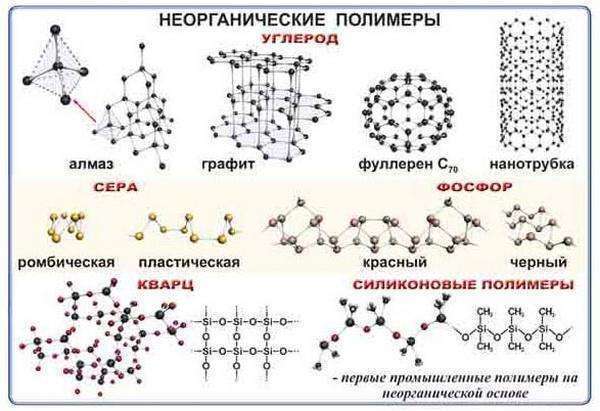
The diamond is highly valued in the jewelry industry. Its hardness has found application in cutting various high strength materials. It is used with a whole stone or simple dust.
When you hear graphite, the first thing that comes to mind is pencils. But the application is wide: mechanical engineering, nuclear industry, etc. Abrasive materials, cutting elements or grinding wheels are few that are made from boron polymer. Iridescent in the rays of the sun, elegant tableware made of rock crystal is the result of synthesis under the influence of a very high temperature of quartz and coal. This is how selenium carbide is used.
Polymers: getting plastics
Materials with a high content of polymers are taken as a basis. The properties of the existing base are being studied, the ability to change its shape from a solid to a liquid or viscous mass and vice versa. Based on the data obtained, the base material refers to thermoplastic polymers or thermoplastic.
Watch a video about what polymers are.
For variety, a filler (it can be solid or gaseous) or an additive can be added to the process. Their synthesis with the base material can significantly extend the life of the product, add color and saturation to its appearance. With the help of a properly selected additional element, it is possible to simplify the production technology, reducing the cost of the finished product.
Solid fillers have a different nature, so compounds with their addition can be recognized:
- asboplasty;
- boroplast;
- graphitoplast;
- organoplasty;
- metal polymer;
- carbon plastic;
- fiberglass.
There are also dispersed-filled joints, where various particles are added, and reinforced ones, where various kinds of fibers are added.
Increasingly large areas of application of polymers force scientists to think about the development of new technologies for their creation, as well as to invent new inorganic compounds.
Polymers, inorganic
If carbon atoms do not participate in the structure of a substance, its chain of molecules, then it is inorganic. Natural inorganic polymers have long been studied and widely used. These include: graphite, ceramics, mica, quartz, diamonds, etc. The most common synthesized ones are silicones.
The chain of this compound is based on silicon and oxygen atoms. Due to the strong bond between atoms, but at the same time flexible, these inorganic polymers acquire the following physical properties:
In everyday life, silicone can be found not only with glue, putties or rubber bands, it is even used in cosmetology.
Polysilane is a little-studied polymer, discovered empirically, but not fully understood. It is also based on the silicon atom, but with the addition of chloride and phenolic compounds. A good conductor of current (although the natural element – copper, is more preferable). Another characteristic feature is heat resistance. Polysilane granules can be heated to very high temperatures. The only thing that can happen in this case is an abrasive.
Empirically created long chains based on tin and germanium. The use of these compounds is not yet widespread, since these metal-based compounds are only being studied.
Another chain based on the silicon atom has been developed. A phosphorus atom and a nitrogen atom were also added to the base. This alternation in the chain has incredible flexibility. Therefore, the resulting polyphosphazene is used using its ability to stretch to incredible sizes and return to its original shape without visible deformation.
Characteristic properties
The characterizing feature of a compound is affected not only by its chemical composition. The molecular weight, polydispersity, degree of polymerization, structure of the atoms that make up its structure are taken into account.